Abstract
Measurable residual disease (MRD) represents the critical population of leukemic cells which persists following anti-leukemic therapy. The presence of MRD has powerful prognostic relevance and represents the reservoir responsible for acute myeloid leukemia (AML) relapse. It is therefore critical to enumerate and molecularly characterize MRD clones in order to better risk stratify AML patients and guide further therapy aimed at preventing relapse and improving outcomes. Current MRD methods, including multi-color flow cytometry (MFC) analysis and bulk NGS sequencing, face challenges in both sensitivity and specificity and are insufficient to distinguish between pre-leukemic and leukemic clones. Consequently, the imperfect predictive value of these techniques hinders meaningful clinical intervention. To overcome these shortcomings associated with conventional methods of MRD detection, we developed a novel single-cell (sc) MRD assay with high sensitivity and specificity by combining precursor/blast population enrichment with integrated scDNA sequencing and immunophenotypic analysis.
We reasoned that the MRD clones responsible for relapse largely reside in immature compartments. Enriched CD34+ and/or CD117+ progenitors from 5 unique patient samples were multiplexed followed by integrated single cell analysis on the MissionBio TapestriV2 scDNA+protein platform. We developed a novel computational approach to deconvolute multiplexed sequencing reads and accurately assign cells to individual patients via K-means clustering and UMAP dimension reduction of germline SNP allele frequencies.
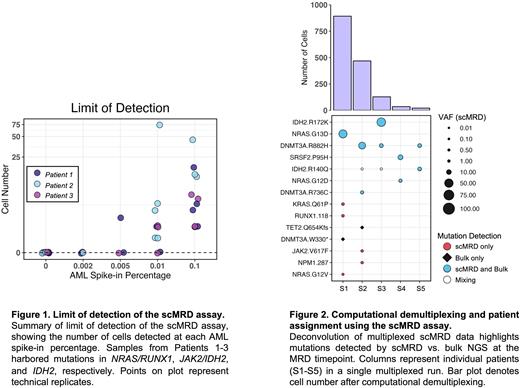
To demonstrate the sensitivity and specificity of our scMRD assay, 10,000, 1,000, 500, and 200 AML blasts from 3 patients harboring known pathologic mutations (NRAS/RUNX1, JAK2/IDH2, and IDH2 alone) were individually spiked into 10 million normal bone marrow cells to test a sensitivity threshold of 0.1%, 0.01%, 0.005%, and 0.002%. Samples were FACS-enriched for blasts, multiplexed, subjected to the TapestriV2 microfluidics platform, and sequenced. The expected pathologic mutations were reliably identified in patients at a threshold of 0.01%, but not in normal subjects, demonstrating that our assay is nearly 10-fold more sensitive than the current ELN MRD standard [Fig 1].
We then applied our assay to 30 banked post-induction MRD samples from 29 patients (median age 71 years old, 15 male/14 female) who had a diagnosis of AML [Fig 2]. Overall, genomic analysis was concordant in 22/30 (73%) samples between bulk NGS (at MRD stage) and scMRD. The mean VAF of mutations was higher by scMRD than bulk NGS, consistent with our hypothesis that mutations are enriched in immature compartments. 17 mutations were identified by scMRD but missed/unreported by bulk NGS, including RUNX1 (n=5), NPM1 (n=3), KRAS (n=2), IDH2 (n=2), WT-1 (n=2), JAK2, TP53 and SRSF2 mutations, 14 of which were present at relapse. Clonal architecture was illustrated by scMRD data with identification of pre-leukemic and leukemic clones. In 5 post-HSCT samples, scMRD readily distinguished between donor vs. host cells and showed higher host cell chimerism compared to conventional STR testing, suggesting that host cells have higher enrichment in immature cell types and represent an early indicator of relapse.
Our scMRD assay also enabled single cell immunophenotypic analysis, and revealed concordant abnormal immunophenotype of leukemic blasts identified by MFC. More importantly, differential immunophenotypic states were identified when comparing pre-leukemic and leukemic clones (i.e. DNMT3A only vs co-mutant DNMT3/NPM1) within and between patients. These data show that integrated genomic/immunophenotypic analysis at the MRD timepoint can distinguish between clonal hematopoiesis and pre-leukemic/leukemic clones with a substantively higher likelihood of relapse.
In conclusion, our scMRD assay not only enhances MRD detection, but also provides a molecular glimpse into the clonal architecture of pre-leukemic/leukemic cells surviving AML therapy. Larger studies are needed to comprehensively elucidate the clonal architecture of MRD and study the dynamics of clonal evolution by comparing clonal architecture and immunophenotype at diagnosis and relapse. Our study paves the road for identifying/eradicating high-risk MRD clones and preventing relapse through a better understanding of the molecular drivers of MRD.
Disclosures
Bowman:Mission Bio: Honoraria, Speakers Bureau. Sciambi:Mission Bio, Inc: Current Employment, Current equity holder in private company, Other: Co-founder & Director of Engineering & Mission Bio, and has a patent pending for a portion of the described work in the submitted abstract.. Llanso:Mission Bio, Inc: Current Employment, Current equity holder in private company, Other: Patent pending for a portion of the described work in the submitted abstract. Goldberg:Moderna, Novavax: Current equity holder in publicly-traded company; Genentech: Consultancy; Aprea, Aptose, AROG, Celularity, Pfizer, Prelude Therapeutics: Research Funding; DAVA Oncology: Honoraria; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees. Dogan:Seattle Genetics: Consultancy; Incyte: Consultancy; Loxo: Consultancy; Peer View: Honoraria; Physicians' Education Resource: Consultancy, Honoraria; Roche: Other: Research Funding; EUSA Pharma: Consultancy; Takeda: Other: Research Funding. Roshal:NGM: Other: Funding; Beat AML: Other: Funding; Auron Therapeutics: Other: Ownership / Equity interests; Provision of services; Celgene: Other: Provision of services; Physicians' Education Resource: Other: Provision of services; Roche: Other: Funding. Levine:Astra Zeneca and Kura: Other: honoraria for invited lectures ; Ajax, Abbvie, Constellation, Zenalis, Celgene, Roche, and Prelude: Other: research support; Qiagen: Other: supervisory board member; Gilead and Novartis: Other: Grant reviews; Imago, Mission Bio, Bakx, Zentalis, Ajax, Auron, Prelude, C4 Therapeutics and Isoplexis: Current equity holder in publicly-traded company, Membership on an entity's Board of Directors or advisory committees; Syndax, Incyte, Janssen, Astellas, Morphosys and Novartis: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.